Update Your Cultivation License With Processing License and Save Massive Costs
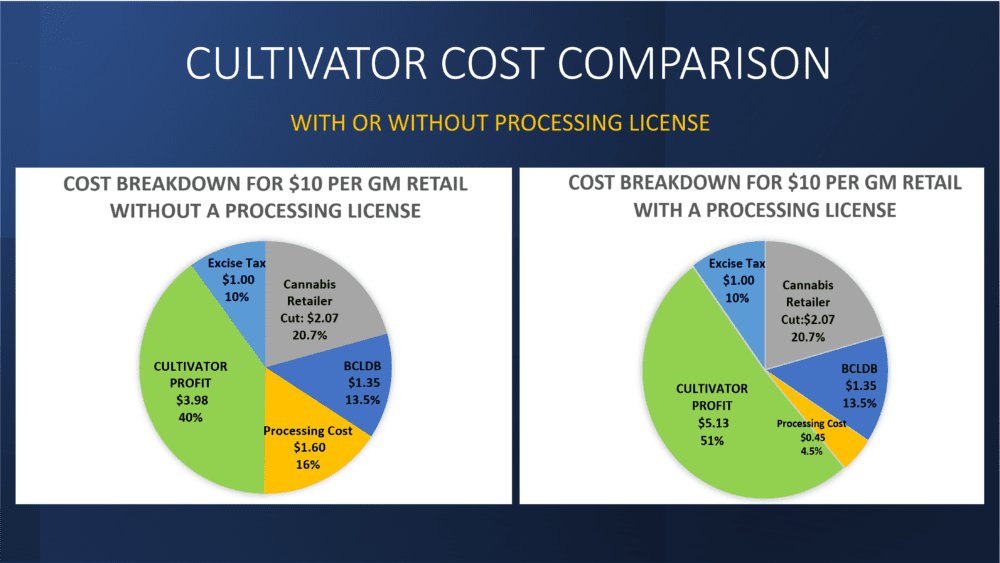
Processing costs for flower products (including pre rolls) run into many hundreds of thousands of dollars for cultivation businesses without processing licences. For example the cost savings on 300 Kgs of flower with a processing license would be $345,000.00
Important Note: Health Canada’s changes to the Cannabis Regulations effective April 19, 2022, means you can request Health Canada approval change to your cannabis license for a processing license and receive a sales license to sell flower at the same time. Previously, a sales amendment was required resulting in several months’ time lag.
This change, in combination with emerging Ontario and BC direct delivery to the retail stores, is a great boon to the industry. Other provinces will follow this precedent, making it essential for cultivators without processing licenses to apply for a processing license to save on processing costs. Also, adding brand recognition and intellectual property value with Trademark registration.
Health Canada’s Processing Licence Holders [1]
The table below (July 5 2022) is an analysis of Health Canada’s published list of licence holders under the Cannabis regulations comprising all cultivation license holders with processing (341) and stand-alone cultivation license holders (340) without processing.

Of the 672 cultivation licenses (July 5 2022), 50% of these do not have processing licenses and cannot sell their flower to the provinces. They either sell their crops to processors at discounted prices, or alternatively pay exorbitant processing fees to sell their flower. The alternative is obvious; request approval change to your cultivation license to process flower products yourself or buy into a processing license though a cooperative such as for example Everest BioPharma international Inc., also providing the opportunity to bypass the province and export internationally.
What are the requirements to request approval change to your cannabis license to obtain the processing license simultaneously with a sales license?
Firstly, the Cannabis Regulations require that a quality assurance person (QAP) be employed. This person is a highly qualified person, with experience in regulatory compliance and standard operating procedures (SOPs), and other qualifications such as a science degree. It is possible to employ a QAP part time, depending on the particular circumstances of the facility.
Secondly, a dedicated space in the facility for the processing activity is required. The floor plan will need to be amended and also the room will have to be modified to meet the requirement of the Cannabis Regulations. Such as for example where positive pressure is needed to prevent cross contamination through air flows into the room.
In addition to these requirements, there are also amendments to the existing documentation such as the original GPP report, the Organizational Security Report, GPP processing SOPs, and record keeping software. A walkthrough video evidencing the new space will need to be filed.
After receipt of the processing license a Notification of New Cannabis Product (NNCP) (60 days) for dried or fresh cannabis is filed with Health Canada, to sell those products into provincial markets, after provincial approval of their SKUs.
[1] Source: Health Canada Website: https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/industry-licencees-applicants/licenced-cultivators-processors-sellers.html.
